A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following condensed structures are branched and which are straight chains? 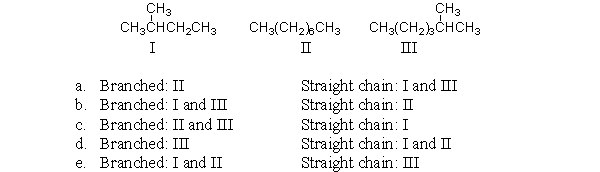
A) a
B) b
C) c
D) d
E) e
G) None of the above
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
Is the following cycloalkane more or less abundant in nature than cyclohexane? 
A) more abundant because it is smaller
B) more abundant because it is more stable
C) less abundant because it is substituted
D) less abundant because the bond angles are not 109.5°
E) more abundant because it is substituted
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A molecule has the molecular formula C5H10. This molecule could be a(n) ______ and a(n) _____.
A) alkane, saturated hydrocarbon
B) alkane, unsaturated hydrocarbon
C) alkene, saturated hydrocarbon
D) alkene, unsaturated hydrocarbon.
E) alkene, alkyne.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the molecules below are branched? 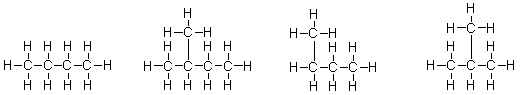 I II III IV
I II III IV
A) I only
B) II and IV
C) II, III, and IV
D) I and III
E) All of these molecules are branched.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these fatty acids is considered a "good fat"? 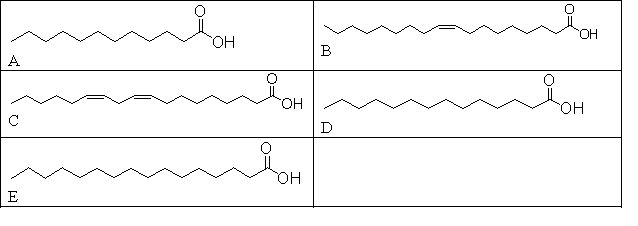
A) None because all fats are bad
B) All of them because all fats taste good
C) B and E because the higher the molecular weight, the better the fat is
D) C and B because these molecules contain cis double bonds
E) A, D, and E because these molecules do not contain any double bonds
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A cyclohexane molecule and a benzene molecule are illustrated below. Which of the four statements describing the relationship between benzene and cyclohexane are true? 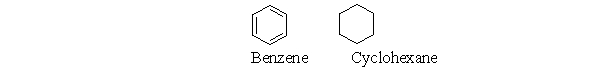 I. Benzene and cyclohexane have different molecular formulas. II. Benzene and cyclohexane are both flat.
III) Benzene and cyclohexane have different C-C bond lengths.
IV) Benzene is more flexible than cyclohexane.
I. Benzene and cyclohexane have different molecular formulas. II. Benzene and cyclohexane are both flat.
III) Benzene and cyclohexane have different C-C bond lengths.
IV) Benzene is more flexible than cyclohexane.
A) Only I is true.
B) I and III are true.
C) I, II, and III are true.
D) I and IV are true.
E) All of the statements are true.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many hydrogens are attached to the carbons indicated below? 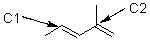
A) C1: 2 C2: 0
B) C1: 0 C2: 2
C) C1: 1 C2: 1
D) C1: 0 C2: 1
E) C1: 1 C2: 0
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following skeletal line structures are isomers? 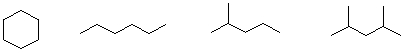 I. II. III. IV.
I. II. III. IV.
A) All of these are isomers.
B) I and II
C) I, II, and III
D) III and IV
E) II and III
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular shape around the carbon indicated with an arrow? 
A) linear
B) trigonal pyramidal
C) tetrahedral
D) bent
E) trigonal planar
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the name of the following hydrocarbon? 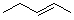
A) trans-3-pentane
B) trans-3-pentene
C) cis-3-pentane
D) trans-2-pentene
E) cis-2-pentene
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which statement best describes the relationship between the following two structures? 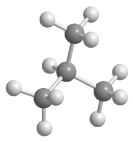
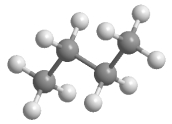
A) They are identical.
B) They have different molecular formulas.
C) They are isomers.
D) They are conformers.
E) One is an alkene and the other is an alkyne.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the IUPAC name of the following hydrocarbon? 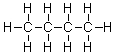
A) propane
B) propene
C) butane
D) pentene
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A compound with the molecular formula CnH2n+2 is called a(n) ______or a ______.
A) alkane, saturated hydrocarbon
B) alkane, unsaturated hydrocarbon
C) alkene, saturated hydrocarbon
D) alkene, unsaturated hydrocarbon
E) alkene, alkyne
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about alkanes is NOT true?
A) Alkanes can move and twist.
B) Merely rotating a C-C bond does not change the identity of an alkane.
C) Alkanes are hydrophilic.
D) Alkanes more rapidly twist and rotate at higher temperatures.
E) Alkanes are often found in a zig-zag shape.
G) C) and E)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
How do IUPAC names differ from the brand names of pharmaceuticals?
A) IUPAC names are systematic and brand names are not.
B) Brand names are systematic and IUPAC names are not.
C) IUPAC names are usually shorter than brand names.
D) Brand names are not used as frequently as IUPAC names.
E) IUPAC names do not follow a set of rules like brand names do.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the parent chain name of the hydrocarbon below? 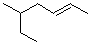
A) butene
B) hexane
C) hexene
D) heptane
E) heptene
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In which of the following choices are the alkenes correctly labeled?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which statement best describes the solubility of hydrocarbons?
A) Hydrocarbons are soluble in water and therefore hydrophobic.
B) Hydrocarbons are insoluble in water and therefore hydrophobic.
C) Hydrocarbons are soluble in water and therefore hydrophilic.
D) Hydrocarbons do not dissolve in any solvent.
E) It is not possible to generalize the solubility of hydrocarbons.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following elements is NOT a heteroatom?
A) carbon
B) sulfur
C) nitrogen
D) oxygen
E) phosphorous
G) A) and B)
Correct Answer

verified
A
Correct Answer
verified
Showing 1 - 20 of 95
Related Exams