A) F
B) C
C) N
D) Al
E) O
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which carbon(s) in the following molecule is (are) sp hybridized? 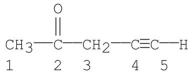
A) carbon 1
B) carbon 2
C) carbons 1,3
D) carbons 4
E) carbons 4,5
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the interaction of two hydrogen 1s atomic orbitals of the same phase.Which of the statements below is an incorrect description of this interaction?
A) A sigma bonding molecular orbital is formed.
B) The molecular orbital formed is lower in energy than a hydrogen 1s atomic orbital.
C) The molecular orbital formed has a node between the atoms.
D) The molecular orbital formed is cylindrically symmetric.
E) A maximum of two electrons may occupy the molecular orbital formed.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many distinct and degenerate p orbitals exist in the second electron shell,where n = 2?
A) 0
B) 1
C) 2
D) 3
E) 4
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Essay
How many nonbonding electron pairs,bonding electron pairs,pi bonds,and sigma bonds are present in CO2?
Correct Answer

verified
4 nonbonding electro...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
The formal charge on nitrogen in the compound below is 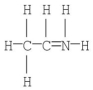
A) +2
B) +1
C) 0
D) -1
E) -2
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Propanal is a compound detected on the surface of comet 67P by the Philae Lander.How many atoms sp2 hybridized atoms are in this molecule? 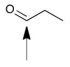
A) 0
B) 1
C) 2
D) 3
E) 4
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Essay
Draw the Lewis structure for CH3N2+.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What orbitals overlap to create the H-C bond in CH3+?
A) sp3-sp3
B) sp2-sp3
C) s-p
D) s-sp2
E) s-sp3
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
Atoms with the same number of protons but different numbers of neutrons are called ________.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following covalent bonds has the largest dipole moment?
A) C-C
B) C-H
C) C-O
D) H-N
E) H-F
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Essay
Give the hybridizations of the carbons,from left to right,in CH3CH  CHCl.
CHCl.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The hydrogen-halogen bond becomes ________ and ________ as the size (atomic weight) of the halogen increases.
A) longer,weaker
B) longer,stronger
C) shorter,weaker
D) shorter,stronger
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Essay
Give the electronic configuration for Ca+2.
Correct Answer

verified
Correct Answer
verified
Short Answer
What kind of molecular orbital (σ,σ*,π,or π*)results when the two atomic orbitals shown below interact in the manner indicated? 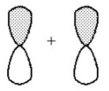
Correct Answer

verified
Correct Answer
verified
Short Answer
What kind of molecular orbital (σ,σ*,π,or π*)results when the two atomic orbitals shown below interact in the manner indicated? 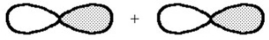
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The N-H single bond in methyl amine (CH3NH2) is a ________ bond formed by the overlap of a(n) ________ orbital on N and a(n) ________ orbital on H.
A) σ; sp2; s
B) σ ; sp3; s
C) π ; sp3; s
D) π ; sp2; p
E) π ; p ; p
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Each lone pair of electrons on the O atom in methanol (CH3OH) occupies a(n) ________ orbital.
A) s
B) p
C) sp
D) sp2
E) sp3
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify the compound with the weakest bond.
A) H2
B) HF
C) HCl
D) HBr
E) HI
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A molecule of acetonitrile CH3CN contains ________ sigma bonds and ________ pi bonds.
A) 5,2
B) 4,3
C) 4,2
D) 2,2
E) 4,0
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 90
Related Exams