A) A-
B) HA
C) H3O+
D) H2A-
E) H2O
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the pH of a 1.8 M solution of HNO3.
A) -0.59
B) -0.26
C) 14.26
D) 13.74
E) 0.26
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If Ka for HCN is 6.2 × 10-10, what is Kb for CN-? 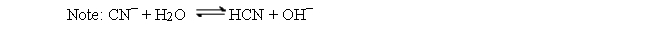
A) 6.2 × 10-24
B) 6.2 × 104
C) 1.6 × 10-5
D) 1.6 × 1023
E) none of these
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For a neutral solution, it must be true that
A) pH = 7.00.
B) [H+] = [OH-].
C) [H2O] = 1 × 10-14
D) [H+] = 0 M.
E) At least two of these must be true.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is true of Arrhenius concept of acids and bases?
A) Acids produce hydroxide ions in aqueous solution and bases produce hydrogen ions.
B) Acids and bases are proton (H+) donors.
C) Acids are proton (H+) acceptors and bases are proton donors.
D) Acids produce hydrogen ions in aqueous solution and bases produce hydroxide ions.
E) Acids are electron donors and bases are electron acceptors.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The pH of a 0.010 M weak acid solution is 5.28. Calculate Ka for this acid.
A) 1.9×10-9
B) 2.8×10-9
C) 5.3×10-6
D) 3.6×10-16
E) 5.3×10-8
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Showing 121 - 126 of 126
Related Exams